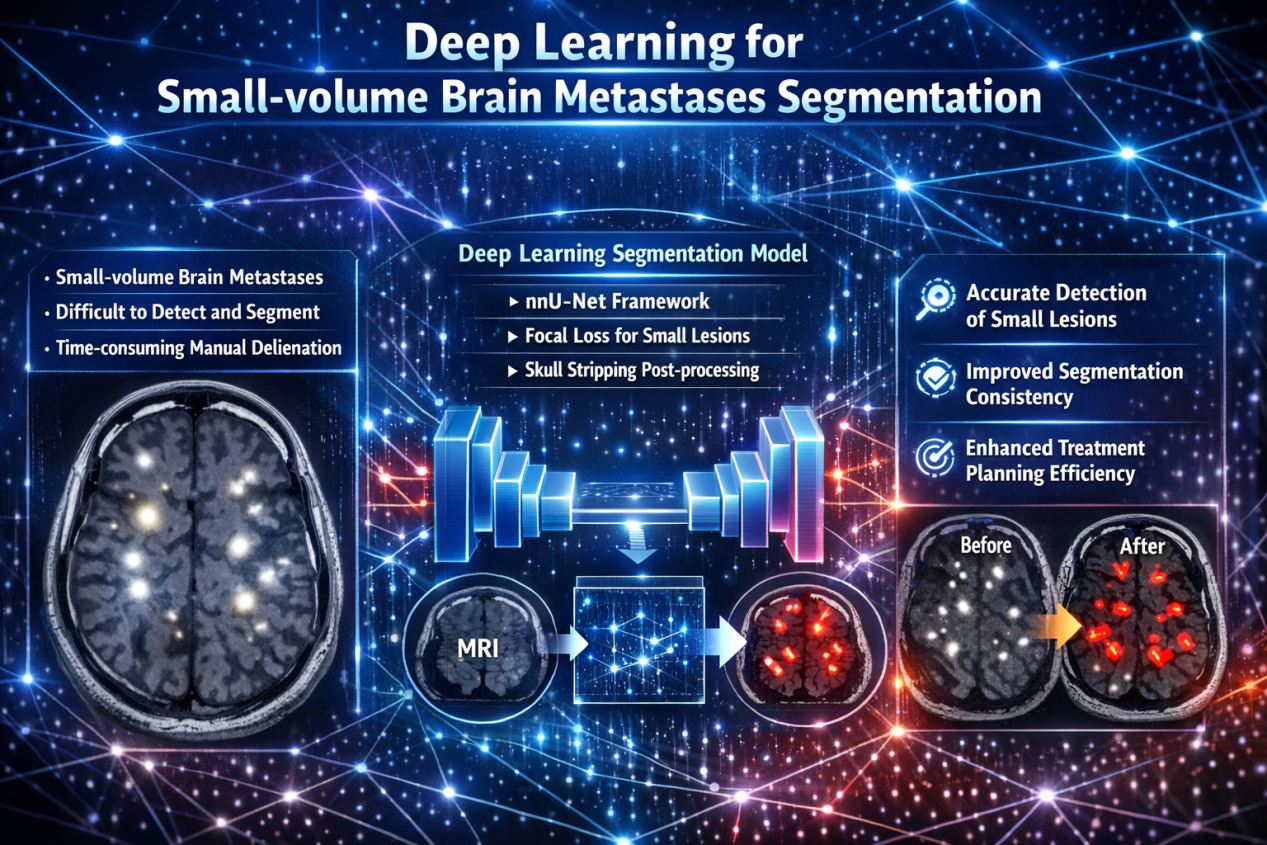
A new study published in Intelligent Oncology presents a deep learning–based approach for the accurate segmentation of small-volume brain metastases in patients with lung cancer, addressing a long-standing challenge in neuro-oncology and radiotherapy planning.
The research was led by June-Goo Lee, the corresponding author, from the University of Ulsan College of Medicine.
Background
Brain metastases are the most common intracranial tumors in adults, frequently arising from primary lung cancer. These lesions are often small, multiple, and widely distributed, making accurate delineation particularly challenging in clinical practice. Precise segmentation is essential for stereotactic radiosurgery (SRS), where even minor inaccuracies can affect treatment efficacy and patient outcomes.
Despite advances in artificial intelligence (AI), most existing models have been developed using datasets dominated by larger lesions or mixed tumor types, limiting their applicability to small-volume metastases.
Study Overview
To address this gap, the investigators developed a deep learning model specifically optimized for small brain metastases originating from lung cancer. The study utilized a large, multi-institutional dataset of 1,413 MRI scans, characterized by a median lesion volume substantially smaller than that of widely used public datasets.
The model was based on the nnU-Net framework and incorporated two key methodological enhancements:
Integration of a focal loss function to improve sensitivity to small lesions;
Application of skull-stripping post-processing to reduce false-positive detections
Model performance was evaluated using both internal and external validation datasets to assess robustness and generalizability.
Key Findings
The proposed model demonstrated reliable and consistent performance:
Internal test set: Dice similarity coefficient of 0.7416;
External validation set: Dice similarity coefficient of 0.7587;
High lesion-level precision (approximately 0.96).
The model also showed strong generalization across different MRI acquisition protocols and scanner manufacturers, supporting its potential applicability in diverse clinical settings.
Clinical Implications
The authors note that this AI-based approach has the potential to:
Reduce the time and variability associated with manual segmentation
Improve accuracy and consistency in target delineation
Enhance efficiency in radiotherapy treatment planning, particularly for SRS
These advances may contribute to improved workflow efficiency and support the broader adoption of precision radiotherapy strategies in patients with metastatic brain disease.
Conclusion
This study demonstrates the feasibility of a dedicated deep learning framework for the segmentation of small-volume brain metastases in lung cancer. By addressing a critical limitation in current AI models, the proposed approach represents a meaningful step toward the integration of automated tools into routine neuro-oncology practice.
Full article available on ScienceDirect:
https://www.sciencedirect.com/science/article/pii/S2950261626000129
Contact Information for Intelligent Oncology:
LinkedIn: @IntelligentOncology
X: @IntelligentOnco
Facebook: @intelligentoncology
Email Address: editorialoffice@intelligent-oncology.net
Official Website: https://www.sciencedirect.com/journal/intelligent-oncology
Submission Link: https://www2.cloud.editorialmanager.com/intonc/default2.aspx
